As Bird Flu In Cows, US Nears Deal on Moderna Vaccine
- Update Time : Thursday, May 30, 2024
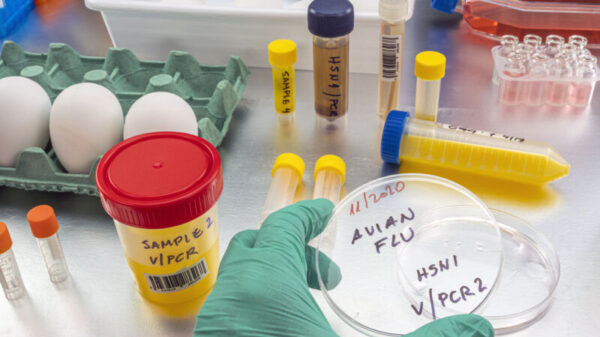
The spread of bird flu in cows is prompting the US to consider funding Moderna’s mRNA H5 vaccine. This move highlights the urgency to combat the threat and protect livestock.
As avian influenza poses risks to both animal and human health, developing a vaccine is crucial for preventing potential outbreaks. Moderna’s innovative mRNA technology offers a promising solution in addressing the evolving nature of flu viruses. By investing in this vaccine, the US aims to strengthen preparedness against emerging diseases and enhance global health security.
The collaboration between government and biopharmaceutical companies underscores the shared responsibility in safeguarding public health and ensuring the well-being of livestock populations.
Bird Flu’s Unusual Jump To Bovines
The zoonotic shift of bird flu to cows has raised concerns about the impacts on cattle health. The spread of bird flu to bovines is a worrying development, with potential consequences for both animal and human health. The emergence of bird flu in cows underscores the need for vaccine development and research into zoonotic diseases. The close proximity to funding Moderna’s mRNA H5 vaccine represents a critical step in addressing the threat posed by bird flu in cattle and potentially humans.

Credit: www.ft.com
Tracking The Outbreak
As bird flu spreads in cows, the US is close to funding Moderna’s mRNA H5 vaccine, marking a significant step in tracking the outbreak. This development brings hope for controlling the spread of the disease and protecting both animals and humans from its impact.
| The bird flu outbreak in cows has raised concerns about the spread of the virus. |
| Geographically, the virus has been detected in multiple regions, prompting widespread alarm. |
| Efforts to contain the outbreak have intensified, with authorities implementing strict measures. |
Economic Ramifications
The spread of bird flu in cows raises concerns in the dairy and beef industries. This outbreak could lead to economic repercussions for farmers and consumers. The global market is closely monitoring the situation and preparing for potential impacts. Farmers may face challenges in maintaining production and meeting consumer demand. Consumers may experience price fluctuations and possible shortages in dairy and beef products. The need for a vaccine is urgent to prevent further spread and protect livestock. The development of a Moderna MRNA H5 vaccine could be a crucial step in controlling the outbreak and stabilizing the market.

Credit: qz.com
Public Health Concerns
Public health concerns arise as bird flu spreads in cows, posing risks for consumers. Human risk assessment is vital to understand the virus’s impact. Monitoring and data collection help gauge the threat. Adherence to safety measures like thorough poultry cooking and good hygiene can reduce exposure.
Moderna’s Vaccine Development
Moderna’s mRNA H5 vaccine has gained significant attention as bird flu spreads among cows. This vaccine holds promise in combating the spread of the disease and has garnered interest from potential funders. The development of Moderna’s vaccine signifies the advancements in technology that have revolutionized the field of medicine.
Efficacy and safety trials are crucial in assessing the vaccine’s effectiveness and ensuring its suitability for widespread use. These trials help determine the vaccine’s ability to protect against the H5 virus strain and its overall safety profile. The success of these trials is essential in gaining regulatory approval and instilling confidence in the vaccine.
Moderna’s mRNA H5 vaccine represents a significant step forward in combating bird flu in cows. The continuous advancements in technology and the rigorous testing process contribute to the development of safe and effective vaccines.
US Government’s Proactive Steps
The US Government is proactively combatting the spread of bird flu in cows through funding Moderna’s mRNA H5 vaccine and strategizing distribution plans. These efforts prioritize public health, minimize transmission risk, and efficiently address emerging health threats in collaboration with pharmaceutical companies. This approach demonstrates the government’s commitment to containing bird flu in livestock and effectively responding to infectious disease challenges.
International Collaborations
International collaborations are essential for the development and distribution of vaccines. Global health organizations play a crucial role in facilitating these partnerships, ensuring that resources and knowledge are shared across borders.
One such example is the current efforts to fund Moderna’s mRNA H5 vaccine to combat the spread of bird flu in cows. This initiative involves multiple countries and organizations working together to support the development and distribution of the vaccine.
| Global Health Organizations Involved: | World Health Organization (WHO) | Coalition for Epidemic Preparedness Innovations (CEPI) | Global Alliance for Vaccines and Immunization (GAVI) | ||
|---|---|---|---|---|---|
| Countries Involved: | United States | Japan | Australia | South Korea | Singapore |
These cross-border partnerships are crucial for responding to global health threats and ensuring that vaccines are accessible to those who need them most.

Credit: www.forbes.com
Future Outlook
As bird flu continues to spread in cows, Moderna’s mRNA H5 vaccine brings hope for the future. To combat this ongoing issue, it is crucial to focus on long-term strategies that prioritize research and innovation.
Investing in research can help us better understand the spread of bird flu and develop more effective vaccines. Innovation is also key in the development of new technologies that can detect and prevent outbreaks before they become widespread.
By prioritizing these long-term strategies, we can not only tackle the current bird flu outbreak but also better prepare for future pandemics. With the funding of Moderna’s mRNA H5 vaccine, we are one step closer to finding a solution to this global health issue.
Frequently Asked Questions
What Is Bird Flu In Cows?
Bird flu in cows is a viral infection caused by the H5N1 avian influenza virus. It can spread from birds to cows and other animals, and even humans who come in contact with infected animals or their products.
What Is Moderna’s Mrna H5 Vaccine?
Moderna’s mRNA H5 vaccine is a vaccine candidate designed to protect against the H5N1 avian influenza virus. It uses messenger RNA (mRNA) technology to instruct cells to produce a protein that triggers an immune response against the virus.
How Close Is The Us To Funding Moderna’s Mrna H5 Vaccine?
The US is reportedly close to funding Moderna’s mRNA H5 vaccine, as the vaccine candidate has shown promising results in preclinical studies. The government has already provided funding for Moderna’s COVID-19 vaccine, which also uses mRNA technology.
Can The Mrna H5 Vaccine Be Used To Prevent Bird Flu In Humans?
While the mRNA H5 vaccine has been designed to protect against the H5N1 avian influenza virus, it has not yet been tested in humans. Clinical trials will be necessary to determine its safety and efficacy in humans.
Conclusion
In light of the concerning spread of bird flu in cows, the potential funding for Moderna’s mRNA H5 vaccine offers hope for combating this serious threat. This advancement reflects a crucial step towards protecting both animal and human populations from the impacts of avian influenza.
Stay informed for more updates.

















Как избежать дубликатов номеров на документах, ознакомьтесь немедленно.
Эффективные способы борьбы с дубликатами номеров, используйте на деле наши советы.
Секреты предотвращения дубликатов номеров, предлагаем изучить.
Какие последствия могут быть при наличии дубликатов номеров, проверьте свои документы уже сегодня.
Советы по правильной нумерации для избежания дубликатов, используйте на деле наши рекомендации.
Почему важно не допускать дубликатов номеров, рекомендуем изучить.
Шаблоны для нумерации без дубликатов, воспользуйтесь этими советами.
Способы детектирования повторений номеров на документах, примените эти советы уже сегодня.
Примеры обнаружения дубликатов номеров в документах, примените указанные методы.
изготовить номера на автомобиль [url=https://dublikat-nomerov777.ru/]изготовить номера на автомобиль[/url] .